All About Glucose


Escrito y verificado por el biólogo Samuel Antonio Sánchez Amador
Glucose is the most abundant organic compound in nature, as it provides energy to the cells of a wide range of living beings. From yeast to humans, this monosaccharide is a fuel that carries out biological processes.
It’s freely present in fruit and honey, but it’s also found in more complex structures where it gives rise to glycogen, chitin, starch, and other polymers of great biological importance. For all these reasons, knowing the chemical characteristics of glucose, its synthesis mechanism, and utility from a nutritional perspective is essential.
The chemical structure of glucose
The first step in finding out all about any molecule in depth is to describe its chemical structure. Fortunately, various portals such as the National Center for Biotechnology Information (NIH) have researched this information for us.
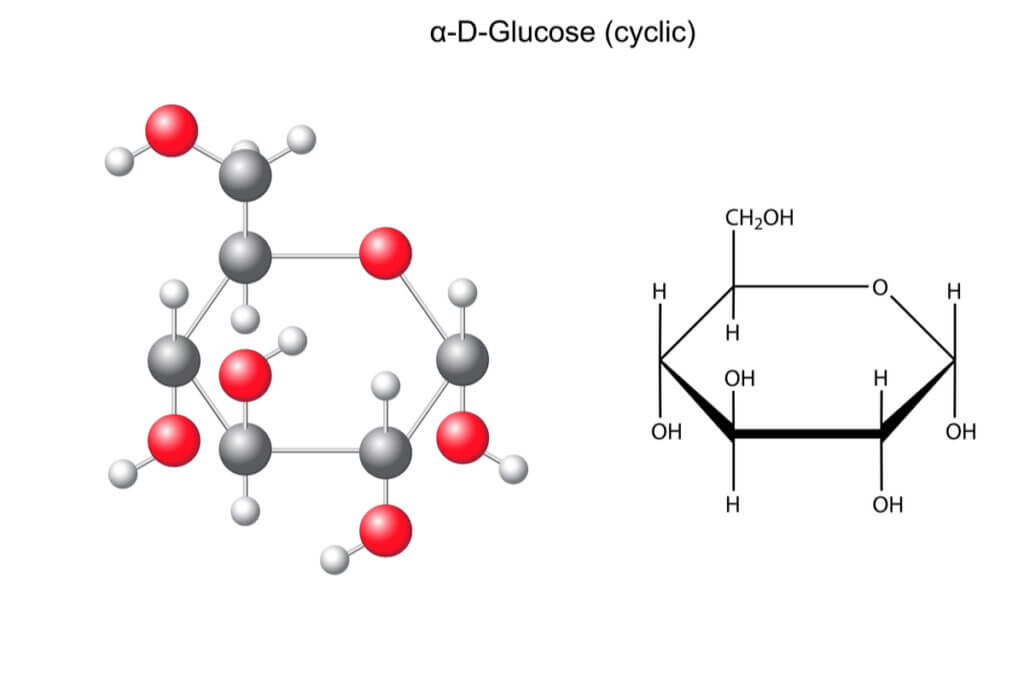
Glucose is a monosaccharide, that is, a sugar that cannot be broken down into other, simpler compounds. Its molecular formula is as follows:
C 6 H 12 O 6
Let’s see what this means. In the first place, we’re dealing with a hexose, because as we can see in its chemical formula, it’s made up of six carbon atoms. Each of these carbons acts as the vertex of its three-dimensional geometric shape, which corresponds to a cyclic ring.
We can also highlight that, in addition, it’s an aldose. This means that it contains a carbonyl (CHO) group at the end of the molecule.
As far as its chemical structure is concerned, it’s also noteworthy that glucose is an isomer of galactose. This means that both molecules have the same chemical formula, but the arrangement of the atoms in space is different. Glucose and galactose are isomers, specifically epimers, as they only differ in the position of carbon number 4.
Glucose biosynthesis
Photosynthetic organisms such as plants are capable of producing glucose through chemical reactions that use inorganic compounds, such as water and carbon dioxide, as a base. These living beings use the energy of the sun to synthesize the molecule.
On the other hand, heterotrophic animals —such as humans— must obtain this monosaccharide through the diet, since we can’t synthesize it ourselves. We can summarize the glucose routes in the following way:
- From the diet and digestion, monosaccharides such as glucose are absorbed, which reach the liver through the portal route.
- In the liver and muscles, it can be obtained from the breakdown of glycogen molecules.
- In the liver and in the renal cortex there’s local production carried out by neoglucogenesis.
Human metabolism
Glucose can be obtained directly from fruits and honey, as monosaccharides after the breakdown of other foods or by going to the glycogen reserves present in the body. In this way, we can see that it has an essential role in different routes:
- Glycogenogenesis: A metabolic pathway that is based on obtaining glycogen from a simpler molecule – glucose-6-phosphate.
- Glycogenolysis: The opposite catabolic process, as it’s about obtaining glucose-6-phosphate from glycogen.
- Glycolysis: The metabolic process by which glucose is oxidized at a cellular level in order to obtain energy.
- Gluconeogenesis (or neoglucogenesis): An anabolic pathway that allows the synthesis of glucose from non-carbohydrate precursors —which aren’t sugars or derivatives—such as various amino acids (lactate, pyruvate, and glycerol).
Finally, it should be noted that monosaccharides such as glucose, galactose or fructose are absorbed in the duodenum and jejunum, both components of the small intestine. They enter intestinal epithelial cells using a sodium (Na+)-dependent cotransport mechanism.
What is glucose used for?
Research highlights that, contrary to popular belief, fat isn’t the main energy storage compound in the human body. 60% of the daily energy for human use comes from carbohydrates, such as starch.
This concept is linked to glucose because these carbohydrates are broken down in the intestine and metabolized, giving rise to glucose, which is stored in the form of glycogen in the liver for times of energy shortage.
In this way, hormones such as insulin or glucagon act by inhibiting or promoting the concentration of glucose in the bloodstream. After food intake, more amounts of insulin are produced, since the storage of glucose in the form of glycogen (gluconeogenesis) is sought.
On the other hand, according to educational portals, glycogenolysis occurs at times of fasting —especially in its second phase— or times when we require high levels of energy in order to obtain fuel immediately.
In this way, a kind of balancing act is produced that regulates the amounts of glucose in the blood effectively. When there’s abundance, glycogen is produced; when shortages are experienced, glucose reserves are degraded.
Diverse functionality
For all these reasons, we can summarize the functions of glucose in three essential pillars:
- Obtaining energy: The oxidation of glucose in the cell cytoplasm (glycolysis) gives rise to ATP, the energy molecule par excellence.
- Reserve: As we have seen, glucose is stored in the form of glucose in the liver and in the muscles. The analog in the plant world is starch, which is found in higher concentrations in fruits, tubers, and roots.
- Structure: Glucose polymers such as cellulose and chitin are an essential structural component in the cell wall of plants and the exoskeleton of many living things.
Glucose and nutrition
As we have been able to see, glucose and nutrition are two interconnected concepts that can’t be differentiated in any way.
It’s one of the two sugars that make up the disaccharides and the basic unit of the polysaccharides already named, such as cellulose and starch. We can get an idea of the nutritional importance of this conglomerate of carbohydrates with the following data:
- A monosaccharide—that is, a simple sugar like glucose—provides 3.74 kilocalories per gram.
- A disaccharide —such as sucrose or lactose— raises this figure to 3.95 kilocalories per gram.
- Finally, the polysaccharide known as starch provides 4.18 kilocalories per gram.
Thus, starch is the main source of energy in the human diet. Again, we remember that this carbohydrate is made up of two glucose polymers: amylose (25%) and amylopectin (75%).
In addition to being part of the most common energy source, it also has certain organoleptic properties. According to sources already cited —and taking sucrose as a reference— glucose has a sweetening power of 0.7. This data is put into perspective when we know that lactose has a sweetening power of 0.25.
Evolutionary significance of glucose and sugars
As a final note, it’s interesting to get to know some of the evolutionary mechanisms that have promoted the production of glucose in fruits and honey.
Before the Triassic period, at some evolutionary moment, various groups of insects passed from a blood-sucking diet —based on the blood of other animals— to a phytophagous one, that is, from the vegetative parts of the plant. This was a severe blow to plant taxa, which found themselves under pressure to form toxic repellent agents for possible predators.
Unfortunately, these toxic substances were energetically very expensive. So, natural selection found a middle ground: if you can’t beat the enemy, join them. Instead of wasting energy producing repellent agents, many plants diverted these metabolic pathways to the formation of sugars such as glucose, expressed in fruits and honey.
The purpose of this is to create an attractive body for living beings, those that prey on certain specific areas, leaving the rest of the plant alone. In addition, they act as a dispersal mechanism for pollen and seeds. It’s no coincidence that fruits are so attractive to us, as nature has helped these products to be sweet, thus dispersing seeds through this medium.
The sugar of life
As we have seen in this article, glucose is a monosaccharide that goes far beyond honey and fruit. Not only is it an excellent source of energy, but it forms part of starch—the most important food in the human diet—and of cellulose or chitin, essential structural compounds.
In addition, it also has various organoleptic and reserve properties. It’s stored in the liver and muscles for times of energy deficiency. It’s quite clear that we’re dealing with a monosaccharide of wide biological and medical interest!
Glucose is the most abundant organic compound in nature, as it provides energy to the cells of a wide range of living beings. From yeast to humans, this monosaccharide is a fuel that carries out biological processes.
It’s freely present in fruit and honey, but it’s also found in more complex structures where it gives rise to glycogen, chitin, starch, and other polymers of great biological importance. For all these reasons, knowing the chemical characteristics of glucose, its synthesis mechanism, and utility from a nutritional perspective is essential.
The chemical structure of glucose
The first step in finding out all about any molecule in depth is to describe its chemical structure. Fortunately, various portals such as the National Center for Biotechnology Information (NIH) have researched this information for us.
Glucose is a monosaccharide, that is, a sugar that cannot be broken down into other, simpler compounds. Its molecular formula is as follows:
C 6 H 12 O 6
Let’s see what this means. In the first place, we’re dealing with a hexose, because as we can see in its chemical formula, it’s made up of six carbon atoms. Each of these carbons acts as the vertex of its three-dimensional geometric shape, which corresponds to a cyclic ring.
We can also highlight that, in addition, it’s an aldose. This means that it contains a carbonyl (CHO) group at the end of the molecule.
As far as its chemical structure is concerned, it’s also noteworthy that glucose is an isomer of galactose. This means that both molecules have the same chemical formula, but the arrangement of the atoms in space is different. Glucose and galactose are isomers, specifically epimers, as they only differ in the position of carbon number 4.

Glucose biosynthesis
Photosynthetic organisms such as plants are capable of producing glucose through chemical reactions that use inorganic compounds, such as water and carbon dioxide, as a base. These living beings use the energy of the sun to synthesize the molecule.
On the other hand, heterotrophic animals —such as humans— must obtain this monosaccharide through the diet, since we can’t synthesize it ourselves. We can summarize the glucose routes in the following way:
- From the diet and digestion, monosaccharides such as glucose are absorbed, which reach the liver through the portal route.
- In the liver and muscles, it can be obtained from the breakdown of glycogen molecules.
- In the liver and in the renal cortex there’s local production carried out by neoglucogenesis.
Human metabolism
Glucose can be obtained directly from fruits and honey, as monosaccharides after the breakdown of other foods or by going to the glycogen reserves present in the body. In this way, we can see that it has an essential role in different routes:
- Glycogenogenesis: A metabolic pathway that is based on obtaining glycogen from a simpler molecule – glucose-6-phosphate.
- Glycogenolysis: The opposite catabolic process, as it’s about obtaining glucose-6-phosphate from glycogen.
- Glycolysis: The metabolic process by which glucose is oxidized at a cellular level in order to obtain energy.
- Gluconeogenesis (or neoglucogenesis): An anabolic pathway that allows the synthesis of glucose from non-carbohydrate precursors —which aren’t sugars or derivatives—such as various amino acids (lactate, pyruvate, and glycerol).
Finally, it should be noted that monosaccharides such as glucose, galactose or fructose are absorbed in the duodenum and jejunum, both components of the small intestine. They enter intestinal epithelial cells using a sodium (Na+)-dependent cotransport mechanism.
What is glucose used for?
Research highlights that, contrary to popular belief, fat isn’t the main energy storage compound in the human body. 60% of the daily energy for human use comes from carbohydrates, such as starch.
This concept is linked to glucose because these carbohydrates are broken down in the intestine and metabolized, giving rise to glucose, which is stored in the form of glycogen in the liver for times of energy shortage.
In this way, hormones such as insulin or glucagon act by inhibiting or promoting the concentration of glucose in the bloodstream. After food intake, more amounts of insulin are produced, since the storage of glucose in the form of glycogen (gluconeogenesis) is sought.
On the other hand, according to educational portals, glycogenolysis occurs at times of fasting —especially in its second phase— or times when we require high levels of energy in order to obtain fuel immediately.
In this way, a kind of balancing act is produced that regulates the amounts of glucose in the blood effectively. When there’s abundance, glycogen is produced; when shortages are experienced, glucose reserves are degraded.
Diverse functionality
For all these reasons, we can summarize the functions of glucose in three essential pillars:
- Obtaining energy: The oxidation of glucose in the cell cytoplasm (glycolysis) gives rise to ATP, the energy molecule par excellence.
- Reserve: As we have seen, glucose is stored in the form of glucose in the liver and in the muscles. The analog in the plant world is starch, which is found in higher concentrations in fruits, tubers, and roots.
- Structure: Glucose polymers such as cellulose and chitin are an essential structural component in the cell wall of plants and the exoskeleton of many living things.
Glucose and nutrition
As we have been able to see, glucose and nutrition are two interconnected concepts that can’t be differentiated in any way.
It’s one of the two sugars that make up the disaccharides and the basic unit of the polysaccharides already named, such as cellulose and starch. We can get an idea of the nutritional importance of this conglomerate of carbohydrates with the following data:
- A monosaccharide—that is, a simple sugar like glucose—provides 3.74 kilocalories per gram.
- A disaccharide —such as sucrose or lactose— raises this figure to 3.95 kilocalories per gram.
- Finally, the polysaccharide known as starch provides 4.18 kilocalories per gram.
Thus, starch is the main source of energy in the human diet. Again, we remember that this carbohydrate is made up of two glucose polymers: amylose (25%) and amylopectin (75%).
In addition to being part of the most common energy source, it also has certain organoleptic properties. According to sources already cited —and taking sucrose as a reference— glucose has a sweetening power of 0.7. This data is put into perspective when we know that lactose has a sweetening power of 0.25.

Evolutionary significance of glucose and sugars
As a final note, it’s interesting to get to know some of the evolutionary mechanisms that have promoted the production of glucose in fruits and honey.
Before the Triassic period, at some evolutionary moment, various groups of insects passed from a blood-sucking diet —based on the blood of other animals— to a phytophagous one, that is, from the vegetative parts of the plant. This was a severe blow to plant taxa, which found themselves under pressure to form toxic repellent agents for possible predators.
Unfortunately, these toxic substances were energetically very expensive. So, natural selection found a middle ground: if you can’t beat the enemy, join them. Instead of wasting energy producing repellent agents, many plants diverted these metabolic pathways to the formation of sugars such as glucose, expressed in fruits and honey.
The purpose of this is to create an attractive body for living beings, those that prey on certain specific areas, leaving the rest of the plant alone. In addition, they act as a dispersal mechanism for pollen and seeds. It’s no coincidence that fruits are so attractive to us, as nature has helped these products to be sweet, thus dispersing seeds through this medium.
The sugar of life
As we have seen in this article, glucose is a monosaccharide that goes far beyond honey and fruit. Not only is it an excellent source of energy, but it forms part of starch—the most important food in the human diet—and of cellulose or chitin, essential structural compounds.
In addition, it also has various organoleptic and reserve properties. It’s stored in the liver and muscles for times of energy deficiency. It’s quite clear that we’re dealing with a monosaccharide of wide biological and medical interest!
- D-glucose, NIH. Recogido a 14 de octubre en https://pubchem.ncbi.nlm.nih.gov/compound/D-Glucose
- rutas de la glucosa, ffis.es. Recogido a 14 de octubre en http://www.ffis.es/volviendoalobasico/3rutas_de_la_glucosa.html
- Home, P. (2004). La glucosa: esa dulce toxina. Diabetes Voice, 49, 5-7.
- Rutas metabólicas del glucógeno, Universidad de Alcalá de Henares (UAH). Recogido a 14 de octubre en http://www3.uah.es/bioquimica/Tejedor/BBM-II_farmacia/R-T8-glucogeno.pdf
- Carbohidratos, manual de nutrición y dietética (UCM). Recogido a 14 de octubre en https://www.ucm.es/data/cont/docs/458-2013-07-24-cap-7-hidratos-carbono.pdf
- Castrejón, V., Carbó, R., & Martínez, M. (2007). Mecanismos moleculares que intervienen en el transporte de la glucosa. Revista de Educación Bioquímica, 26(2), 49-57.
- Fontúrbel, F. (2002). Rol de la coevolución planta–insecto en la evolución de las flores cíclicas en las angiospermas. Ciencia Abierta, 17(11).
Este texto se ofrece únicamente con propósitos informativos y no reemplaza la consulta con un profesional. Ante dudas, consulta a tu especialista.







