Hypoglycemic Drugs: What Types Are There?

Diabetes mellitus (DM) is a carbohydrate metabolism disorder characterized by chronic hyperglycemia. The pathophysiology of this disease describes a deficit in insulin secretion and/or activity. It’s usually treated with hypoglycemic drugs.
Within this disease there are mainly two types:
- Type 1 DM: This appears at an early age, the patient has destruction of pancreatic β cells, which are the cells that secrete insulin, and therefore these people depend on the administration of exogenous insulin to survive.
- Type 2 DM: This type of diabetes appears at an older age, has a much higher prevalence, and can develop from various causes.
In the treatment of this disease, a specific diet and regular physical exercise should also be considered.

Hypoglycemic drugs
They are drugs that, through different mechanisms, manage to lower blood glucose levels.
Insulin
This is the hypoglycemic agent par excellence, and the human body is able to synthesize it naturally in pancreatic β cells. Insulin secretion is triggered mainly by the stimulus that causes the increase in blood glucose levels.
Insulin is an anabolic hormone that favors the uptake, utilization, and storage of glucose, amino acids, and lipids after eating. It also inhibits catabolic processes such as the breakdown of glycogen, fats, and proteins.
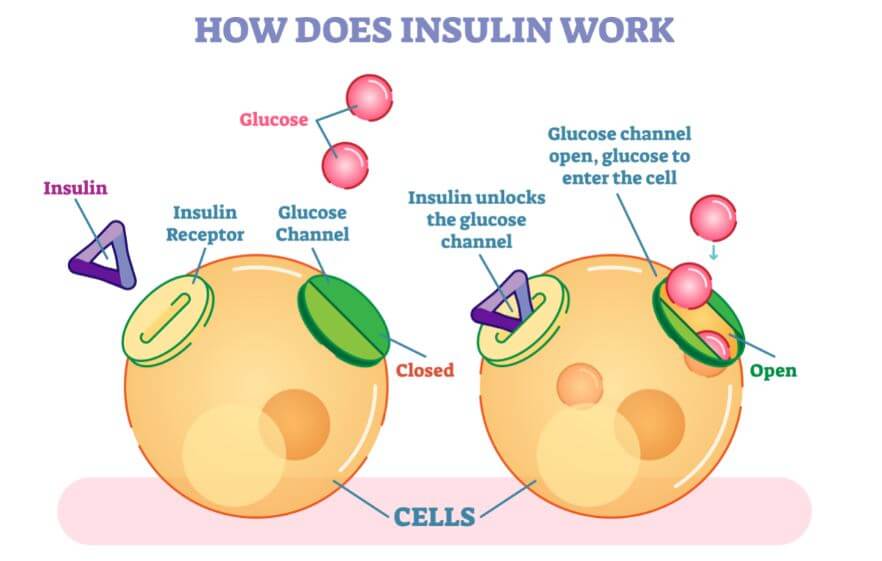
This hormone reduces blood glucose levels as a result of increased glucose uptake by the tissues, and decreased glucose release from the liver.
To achieve peripheral glucose uptake by adipose and muscle tissue, insulin activates the cellular transporter GLUT4, which causes the entry of glucose and its storage in the cells of these tissues.
All insulins marketed are practically the same as the original hormone that the human body synthesizes naturally, but modifications have been made to achieve different kinetics in each of them.
Insulin types
- Regular insulin: This is the only one that can be administered intramuscularly or intravenously in addition to the subcutaneous route, which is the most widely used. It’s fast acting and should be administered 15-30 minutes before meals.
- Lis-Pro and Aspart: These are human insulin analogs and have ultrashort action, so they should be administered immediately before meals.
- Wide-acting insulin: These are used to achieve basal insulin levels. They should be injected 30-45 minutes before food intake or at bedtime.
- NPH: Neutral insulin associated with protamine crystals.
- Slow insulin: A delay in absorption and prolonged activity are achieved thanks to the suspension with zinc crystals.
- Ultra-slow insulin: This preparation has a prolonged onset of action and a broad effect.
- Insulin glargine: Similar to ultra slow. It maintains basal levels of insulin in the blood and is administered only once a day.
Oral hypoglycemic
Sulfonylureas
Sulfonylureas exert a stimulating effect on insulin secretion thanks to their binding with the SUR membrane receptor that induces a blockage of K channels in pancreatic β cells, which produces a depolarization and as a consequence, an increase in the entry Ca² † that triggers insulin secretion.
In addition, these drugs can raise insulin levels by reducing its liver clearance. These drugs have good oral absorption, achieving maximum plasma concentrations in 2-4 hours.
The most commonly used drugs in this group are glimepiride, glibenclamide, and gliclazide. As they’re insulinotropic drugs, that is, they increase insulin secretion, sulfonylureas are recommended to treat type 2 DM except in cases where there’s a problem in insulin secretion.
The frequency with which adverse reactions appear when taking these drugs is very low and they’re usually mild. The most common is hypoglycemia followed by skin hypersensitivity.
Meglitinide analogs
Within this group, we highlight two drugs: repaglinide and nateglinide. These drugs act through the same mechanism described in the previous group. The advantage they have is that they’re absorbed and begin to exert their effect quicker than sulfonylureas; they achieve their action in 30 minutes.
These drugs are recommended in patients with type 2 DM with severe postprandial hyperglycemia.
The risk of hypoglycemia as an adverse effect is significantly reduced with these drugs compared to sulfonylureas. They can lead to an increase in transaminases that would require regular liver function tests.
Biguanides
Metformin is the most widely used agent within this group of drugs. This drug lowers blood glucose levels without acting on pancreatic β cells.
Its effect is due to the fact that they reduce hepatic gluconeogenesis (hepatic synthesis of glucose), reduce glycogenolysis (breakdown of stored glycogen molecules to obtain glucose) to a lesser degree, potentiate the effects of insulin in adipose and muscle tissue, and decrease the intestinal glucose absorption.
Biguanides are recommended in the treatment of type 2 DM that doesn’t improve with diet or exercise. It’s also often prescribed when this disease appears associated with obesity. The oral bioavailability of these drugs is around 50-60% and the kidney actively excretes them.
A great advantage of biguanides is that they don’t pose a risk of hypoglycemia since they don’t stimulate insulin secretion. The most commonly seen adverse effects are bloating, nausea, and vomiting.
Α-glucosylase inhibitors
Within this group, acarbose and miglitol are used. They act by inhibiting α-glucosylases at an intestinal level, thus delaying the transformation of complex carbohydrates into simple ones, thus hindering their absorption.
The main problem with the use of these drugs is that complex carbohydrates reach the large intestine where they ferment, causing diarrhea and flatulence.

Acarbose is virtually not absorbed and is broken down by intestinal bacteria. Miglitol is well absorbed and is eliminated by the kidneys without undergoing modifications. It’s also excreted through breast milk, which is why it’s contraindicated during lactation.
They’re often prescribed in type 2 DM alone or in association with other hypoglycemic agents. They’re especially useful in patients with severe postprandial hyperglycemia.
Apart from the adverse effects discussed above, significant increases in transaminase levels have also been observed with its use at high doses. In monotherapy they don’t produce hypoglycemia.
Glitazones
Glitazones are PPARγ receptor agonists and reduce basal and postprandial blood glucose by stimulating peripheral glucose uptake, inhibiting hepatic gluconeogenesis, and stimulating glycogen accumulation.
They also increase the synthesis of the GLUT4 transporter and its translocation to the membrane in both adipose and muscle tissue. These drugs are indicated in type 2 DM and are usually used in conjunction with other treatments. The best known are rosiglitazone and pioglitazone.
Dipeptyl peptidase-4 inhibitors
They work by inhibiting the enzyme dipeptidyl peptidase-4, which is responsible for destroying GLP-1, which is an incretin that stimulates insulin secretion. Sitagliptin and vidagliptin are two of the drugs in this group.
They have a low risk of causing hypoglycemia and are often used in combination with other drugs.
GLP-1 receptor analogs

These drugs act by mimicking the action of GLP-1 but the duration of their effect is much longer, which is why they achieve a longer increase in insulin secretion. They’re administered subcutaneously and include liraglutide and exenatide.
SGTL-2 inhibitors
SGTL-2 is a renal sodium and glucose cotransporter. These drugs owe their pharmacological action to the inhibition of these receptors and therefore the reabsorption of glucose at the nephron level. As a consequence, glucose is expelled through the urine.
The most widely used is dapagliflozin. Due to a greater presence of glucose in the urine, the consumption of these drugs can accentuate the presence of urinary tract infections. In turn, it isn’t recommended in patients with kidney problems.
Diabetes mellitus (DM) is a carbohydrate metabolism disorder characterized by chronic hyperglycemia. The pathophysiology of this disease describes a deficit in insulin secretion and/or activity. It’s usually treated with hypoglycemic drugs.
Within this disease there are mainly two types:
- Type 1 DM: This appears at an early age, the patient has destruction of pancreatic β cells, which are the cells that secrete insulin, and therefore these people depend on the administration of exogenous insulin to survive.
- Type 2 DM: This type of diabetes appears at an older age, has a much higher prevalence, and can develop from various causes.
In the treatment of this disease, a specific diet and regular physical exercise should also be considered.

Hypoglycemic drugs
They are drugs that, through different mechanisms, manage to lower blood glucose levels.
Insulin
This is the hypoglycemic agent par excellence, and the human body is able to synthesize it naturally in pancreatic β cells. Insulin secretion is triggered mainly by the stimulus that causes the increase in blood glucose levels.
Insulin is an anabolic hormone that favors the uptake, utilization, and storage of glucose, amino acids, and lipids after eating. It also inhibits catabolic processes such as the breakdown of glycogen, fats, and proteins.

This hormone reduces blood glucose levels as a result of increased glucose uptake by the tissues, and decreased glucose release from the liver.
To achieve peripheral glucose uptake by adipose and muscle tissue, insulin activates the cellular transporter GLUT4, which causes the entry of glucose and its storage in the cells of these tissues.
All insulins marketed are practically the same as the original hormone that the human body synthesizes naturally, but modifications have been made to achieve different kinetics in each of them.
Insulin types
- Regular insulin: This is the only one that can be administered intramuscularly or intravenously in addition to the subcutaneous route, which is the most widely used. It’s fast acting and should be administered 15-30 minutes before meals.
- Lis-Pro and Aspart: These are human insulin analogs and have ultrashort action, so they should be administered immediately before meals.
- Wide-acting insulin: These are used to achieve basal insulin levels. They should be injected 30-45 minutes before food intake or at bedtime.
- NPH: Neutral insulin associated with protamine crystals.
- Slow insulin: A delay in absorption and prolonged activity are achieved thanks to the suspension with zinc crystals.
- Ultra-slow insulin: This preparation has a prolonged onset of action and a broad effect.
- Insulin glargine: Similar to ultra slow. It maintains basal levels of insulin in the blood and is administered only once a day.

Oral hypoglycemic
Sulfonylureas
Sulfonylureas exert a stimulating effect on insulin secretion thanks to their binding with the SUR membrane receptor that induces a blockage of K channels in pancreatic β cells, which produces a depolarization and as a consequence, an increase in the entry Ca² † that triggers insulin secretion.
In addition, these drugs can raise insulin levels by reducing its liver clearance. These drugs have good oral absorption, achieving maximum plasma concentrations in 2-4 hours.
The most commonly used drugs in this group are glimepiride, glibenclamide, and gliclazide. As they’re insulinotropic drugs, that is, they increase insulin secretion, sulfonylureas are recommended to treat type 2 DM except in cases where there’s a problem in insulin secretion.
The frequency with which adverse reactions appear when taking these drugs is very low and they’re usually mild. The most common is hypoglycemia followed by skin hypersensitivity.
Meglitinide analogs

Within this group, we highlight two drugs: repaglinide and nateglinide. These drugs act through the same mechanism described in the previous group. The advantage they have is that they’re absorbed and begin to exert their effect quicker than sulfonylureas; they achieve their action in 30 minutes.
These drugs are recommended in patients with type 2 DM with severe postprandial hyperglycemia.
The risk of hypoglycemia as an adverse effect is significantly reduced with these drugs compared to sulfonylureas. They can lead to an increase in transaminases that would require regular liver function tests.
Biguanides

Metformin is the most widely used agent within this group of drugs. This drug lowers blood glucose levels without acting on pancreatic β cells.
Its effect is due to the fact that they reduce hepatic gluconeogenesis (hepatic synthesis of glucose), reduce glycogenolysis (breakdown of stored glycogen molecules to obtain glucose) to a lesser degree, potentiate the effects of insulin in adipose and muscle tissue, and decrease the intestinal glucose absorption.
Biguanides are recommended in the treatment of type 2 DM that doesn’t improve with diet or exercise. It’s also often prescribed when this disease appears associated with obesity. The oral bioavailability of these drugs is around 50-60% and the kidney actively excretes them.
A great advantage of biguanides is that they don’t pose a risk of hypoglycemia since they don’t stimulate insulin secretion. The most commonly seen adverse effects are bloating, nausea, and vomiting.
Α-glucosylase inhibitors
Within this group, acarbose and miglitol are used. They act by inhibiting α-glucosylases at an intestinal level, thus delaying the transformation of complex carbohydrates into simple ones, thus hindering their absorption.
The main problem with the use of these drugs is that complex carbohydrates reach the large intestine where they ferment, causing diarrhea and flatulence.

Acarbose is virtually not absorbed and is broken down by intestinal bacteria. Miglitol is well absorbed and is eliminated by the kidneys without undergoing modifications. It’s also excreted through breast milk, which is why it’s contraindicated during lactation.
They’re often prescribed in type 2 DM alone or in association with other hypoglycemic agents. They’re especially useful in patients with severe postprandial hyperglycemia.
Apart from the adverse effects discussed above, significant increases in transaminase levels have also been observed with its use at high doses. In monotherapy they don’t produce hypoglycemia.
Glitazones
Glitazones are PPARγ receptor agonists and reduce basal and postprandial blood glucose by stimulating peripheral glucose uptake, inhibiting hepatic gluconeogenesis, and stimulating glycogen accumulation.
They also increase the synthesis of the GLUT4 transporter and its translocation to the membrane in both adipose and muscle tissue. These drugs are indicated in type 2 DM and are usually used in conjunction with other treatments. The best known are rosiglitazone and pioglitazone.
Dipeptyl peptidase-4 inhibitors
They work by inhibiting the enzyme dipeptidyl peptidase-4, which is responsible for destroying GLP-1, which is an incretin that stimulates insulin secretion. Sitagliptin and vidagliptin are two of the drugs in this group.
They have a low risk of causing hypoglycemia and are often used in combination with other drugs.
GLP-1 receptor analogs

These drugs act by mimicking the action of GLP-1 but the duration of their effect is much longer, which is why they achieve a longer increase in insulin secretion. They’re administered subcutaneously and include liraglutide and exenatide.
SGTL-2 inhibitors
SGTL-2 is a renal sodium and glucose cotransporter. These drugs owe their pharmacological action to the inhibition of these receptors and therefore the reabsorption of glucose at the nephron level. As a consequence, glucose is expelled through the urine.
The most widely used is dapagliflozin. Due to a greater presence of glucose in the urine, the consumption of these drugs can accentuate the presence of urinary tract infections. In turn, it isn’t recommended in patients with kidney problems.
- Velélazquez: Farmacología Básica y Clínica. Lorenzo P., Moreno A., Leza J.C., Lizasoain I., Moro M.A.
- Inhibidores de la dipeptidil peptidasa-4: farmacodinamia, farmacocinética y seguridad Di Girolamo G., Tamez Peña A.L., Tamez Pérez H.E.,
Este texto se ofrece únicamente con propósitos informativos y no reemplaza la consulta con un profesional. Ante dudas, consulta a tu especialista.







