What Is Dopamine?

Escrito y verificado por la psicóloga Paula Villasante
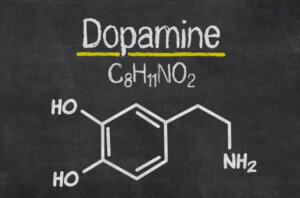
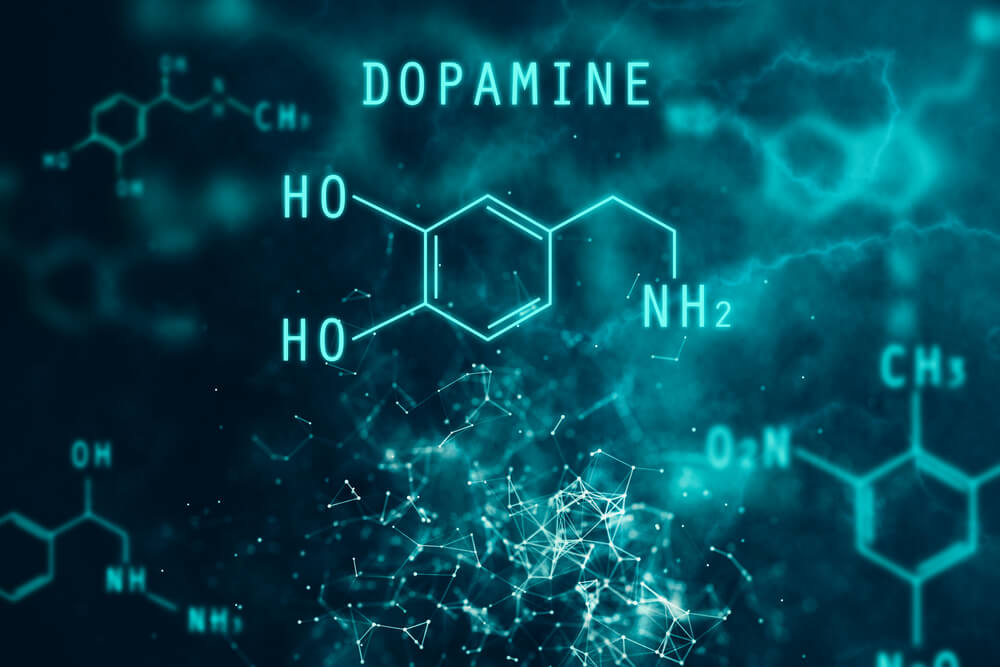
Dopamine is the most important catecholaminergic neurotransmitter in the central nervous system (CNS). This neurotransmitter participates in the regulation of various functions such as motor behavior, emotion, and affectivity, as well as in neuroendocrine communication (1).
Dopamine is synthesized from the amino acid L-tyrosine. In addition, there are mechanisms that very precisely regulate its synthesis and release.
Thus, molecular cloning techniques have allowed the identification of 5 types of dopamine receptors, all of them coupled to G proteins and divided into two pharmacological families called D1 and D2.
Dopaminergic systems
Dopaminergic neurons and their projections can be grouped into 3 main systems (2) :
Ultrashort systems
There are two ultrashort systems. The first is formed by the dopaminergic cells of the olfactory bulb. Meanwhile, the second system is made up of inter-plexiform neurons (similar to amacrines) present between the inner and outer plexiform layers of the retina.
Intermediate length systems
Mid-length systems include:
a) The tuberoinfudibular system, originating from the dopaminergic cells located in the arcuate and periventricular hypothalamic nuclei.
b) Neurons located in the dorsal and posterior hypothalamus. These send projections to the anterior dorsal hypothalamus and to the septolateral nuclei.
c) The medullary periventricular group, which includes dopaminergic neurons located on the periphery of the nuclei of the solitary and dorsal motor tract of the vagus nerve. It also includes scattered cells in the tegmental prolongation of the periaqueductal gray matter.
Long systems
This group includes neurons in the retrorubral region, the ventral tegmental area, and the substantia nigra compact. They send projections to three main regions:
- The neostriatum (caudate nuclei and putamen).
- The limbic cortex (entorhinal, medial prefrontal, and cingulum).
- Other limbic structures (such as the septum, olfactory tubercle, nucleus accumens, amygdala, and piriform cortex). Within this group, there are also two of the most important dopaminergic pathways. These are the nigrostriatal pathway and the mesolimbic pathway.
Synthesis of dopamine
The synthesis of the neurotransmitter takes place in the dopaminergic nerve terminals where the responsible enzymes, tyrosine hydroxylase (TH) and aromatic amino acid decarboxylase or L-DOPA decarboxylase are found in high concentration (2, 3).
The works of Nagatsu et al. (4) and Levitt et al. (5) demonstrated that the hydroxylation of the amino acid L-tyrosine is the point of regulation of catecholamine synthesis in the CNS. Thus, consequently, TH is the limiting enzyme for the synthesis of dopamine, norepinephrine, and epinephrine.
TH is a 498 amino acid peptide present predominantly in the cytosolic fraction of the catecholaminergic terminals (6).
Dopamine release
At the dopaminergic terminals, the neurotransmitter is synthesized in the cytoplasm from where it can be released directly into the synaptic space or be transported into the synaptic vesicles to be released by exocytosis.
Release by exocytosis
In this process, the dopamine contained in vesicles is released to the outside when the vesicular membrane fuses with the membrane of the presynaptic terminal. This mechanism is made up of several stages.
First, the vesicles transport the neurotransmitter inside them through a carrier protein with 12 transmembrane domains that uses an electrochemical gradient generated by a proton (H +) pump (ATPase) (7).
Independent release of calcium
This second type of dopamine release is characteristically inhibited by drugs that block the dopamine transporter present in the synaptic terminal membrane.
The function of this transporter is to terminate the action of the neurotransmitter, capturing it in the terminal. Thus, under certain conditions, the transporter operates in the opposite direction, releasing dopamine to the outside (8).
Dopamine synthesis and release
The dopamine that is preferentially released in response to synaptic stimulation has been found to be newly synthesized.
The neurotransmitter thus appears to be found in two metabolic pools, both vesicular; one that contains newly synthesized dopamine and a second that would correspond to a pool that functions as a store (9).
Regulation of dopamine release
Regulation of dopamine release can occur through two types of regulation. These are:
- Regulation by autoreceptors
- Regulation by heteroreceptors
Dopamine receptors
Currently, receptors are defined as molecules or molecular compositions that can selectively recognize a ligand (agonist or antagonist) and be activated by the ligand with intrinsic efficiency (agonist) to initiate a cellular event (10).
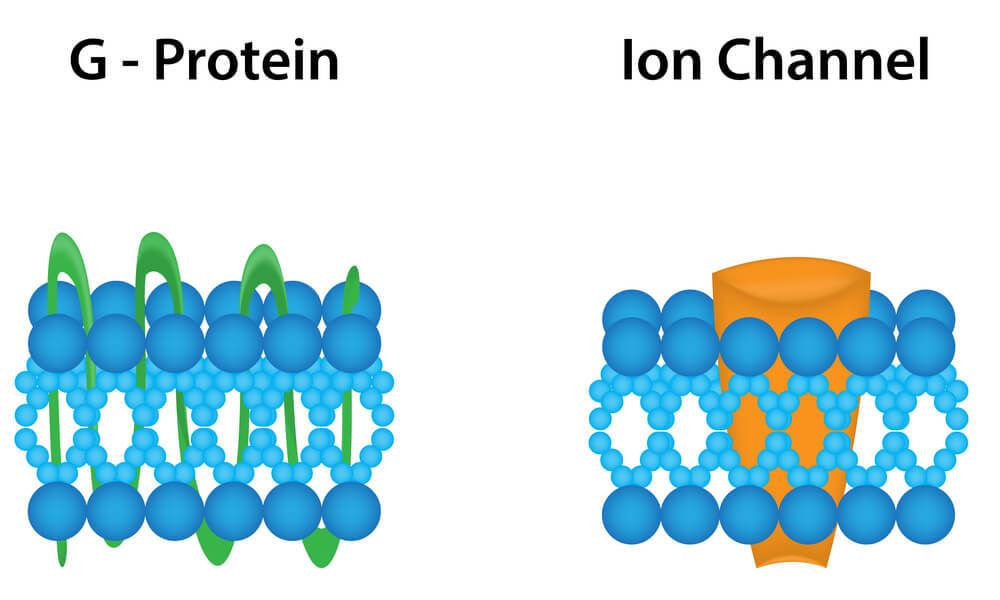
Receptors for dopamine belong to the superfamily of receptors (with more than 100 members) coupled to G proteins.
In this family of receptors, the neurotransmitter recognition and the effector molecule (typically an enzyme that produces a diffusible second messenger) are different entities, coupled together by a protein with the ability to bind guanine nucleotides (G protein) (11).
Dopamine is a very important neurotransmitter in the central nervous system. The functional effects of dopamine are exerted through the activation of 5 receptor subtypes, all of them coupled to G proteins and grouped into two pharmacological families, D1 (D1 and D5) and D2 (D2, D3, D4).
Various alterations of the CNS (Parkinson’s disease, schizophrenia and drug addiction, among others) have been linked to disorders of dopamine transmission.
Consequently, the study of aspects that haven’t yet been understood regarding the function of dopamine, of the different receptor subtypes, and of the mechanisms of signal transduction, will not only allow us to advance in the understanding of how the dopamine systems work, but also to design new pharmacological strategies that affect the therapy of these pathological processes (1).
Dopamine is the most important catecholaminergic neurotransmitter in the central nervous system (CNS). This neurotransmitter participates in the regulation of various functions such as motor behavior, emotion, and affectivity, as well as in neuroendocrine communication (1).
Dopamine is synthesized from the amino acid L-tyrosine. In addition, there are mechanisms that very precisely regulate its synthesis and release.
Thus, molecular cloning techniques have allowed the identification of 5 types of dopamine receptors, all of them coupled to G proteins and divided into two pharmacological families called D1 and D2.
Dopaminergic systems

Dopaminergic neurons and their projections can be grouped into 3 main systems (2) :
Ultrashort systems
There are two ultrashort systems. The first is formed by the dopaminergic cells of the olfactory bulb. Meanwhile, the second system is made up of inter-plexiform neurons (similar to amacrines) present between the inner and outer plexiform layers of the retina.
Intermediate length systems
Mid-length systems include:
a) The tuberoinfudibular system, originating from the dopaminergic cells located in the arcuate and periventricular hypothalamic nuclei.
b) Neurons located in the dorsal and posterior hypothalamus. These send projections to the anterior dorsal hypothalamus and to the septolateral nuclei.
c) The medullary periventricular group, which includes dopaminergic neurons located on the periphery of the nuclei of the solitary and dorsal motor tract of the vagus nerve. It also includes scattered cells in the tegmental prolongation of the periaqueductal gray matter.
Long systems
This group includes neurons in the retrorubral region, the ventral tegmental area, and the substantia nigra compact. They send projections to three main regions:
- The neostriatum (caudate nuclei and putamen).
- The limbic cortex (entorhinal, medial prefrontal, and cingulum).
- Other limbic structures (such as the septum, olfactory tubercle, nucleus accumens, amygdala, and piriform cortex). Within this group, there are also two of the most important dopaminergic pathways. These are the nigrostriatal pathway and the mesolimbic pathway.
Synthesis of dopamine

The synthesis of the neurotransmitter takes place in the dopaminergic nerve terminals where the responsible enzymes, tyrosine hydroxylase (TH) and aromatic amino acid decarboxylase or L-DOPA decarboxylase are found in high concentration (2, 3).
The works of Nagatsu et al. (4) and Levitt et al. (5) demonstrated that the hydroxylation of the amino acid L-tyrosine is the point of regulation of catecholamine synthesis in the CNS. Thus, consequently, TH is the limiting enzyme for the synthesis of dopamine, norepinephrine, and epinephrine.
TH is a 498 amino acid peptide present predominantly in the cytosolic fraction of the catecholaminergic terminals (6).
Dopamine release
At the dopaminergic terminals, the neurotransmitter is synthesized in the cytoplasm from where it can be released directly into the synaptic space or be transported into the synaptic vesicles to be released by exocytosis.
Release by exocytosis
In this process, the dopamine contained in vesicles is released to the outside when the vesicular membrane fuses with the membrane of the presynaptic terminal. This mechanism is made up of several stages.
First, the vesicles transport the neurotransmitter inside them through a carrier protein with 12 transmembrane domains that uses an electrochemical gradient generated by a proton (H +) pump (ATPase) (7).
Independent release of calcium

This second type of dopamine release is characteristically inhibited by drugs that block the dopamine transporter present in the synaptic terminal membrane.
The function of this transporter is to terminate the action of the neurotransmitter, capturing it in the terminal. Thus, under certain conditions, the transporter operates in the opposite direction, releasing dopamine to the outside (8).
Dopamine synthesis and release
The dopamine that is preferentially released in response to synaptic stimulation has been found to be newly synthesized.
The neurotransmitter thus appears to be found in two metabolic pools, both vesicular; one that contains newly synthesized dopamine and a second that would correspond to a pool that functions as a store (9).
Regulation of dopamine release
Regulation of dopamine release can occur through two types of regulation. These are:
- Regulation by autoreceptors
- Regulation by heteroreceptors
Dopamine receptors

Currently, receptors are defined as molecules or molecular compositions that can selectively recognize a ligand (agonist or antagonist) and be activated by the ligand with intrinsic efficiency (agonist) to initiate a cellular event (10).
Receptors for dopamine belong to the superfamily of receptors (with more than 100 members) coupled to G proteins.
In this family of receptors, the neurotransmitter recognition and the effector molecule (typically an enzyme that produces a diffusible second messenger) are different entities, coupled together by a protein with the ability to bind guanine nucleotides (G protein) (11).
Dopamine is a very important neurotransmitter in the central nervous system. The functional effects of dopamine are exerted through the activation of 5 receptor subtypes, all of them coupled to G proteins and grouped into two pharmacological families, D1 (D1 and D5) and D2 (D2, D3, D4).
Various alterations of the CNS (Parkinson’s disease, schizophrenia and drug addiction, among others) have been linked to disorders of dopamine transmission.
Consequently, the study of aspects that haven’t yet been understood regarding the function of dopamine, of the different receptor subtypes, and of the mechanisms of signal transduction, will not only allow us to advance in the understanding of how the dopamine systems work, but also to design new pharmacological strategies that affect the therapy of these pathological processes (1).
-
Bahena-Trujillo, R., Flores, G., & Arias-Montaño, J. A. (2000). Dopamina: síntesis, liberación y receptores en el Sistema Nervioso Central. Revista Biomédica, 11(1), 39-60.
-
Cooper, J. R., Bloom, F. E., & Roth, R. H. (2003). The biochemical basis of neuropharmacology. Oxford University Press, USA.
-
Freund, T. F., Powell, J. F., & Smith, A. D. (1984). Tyrosine hydroxylase-immunoreactive boutons in synaptic contact with identified striatonigral neurons, with particular reference to dendritic spines. Neuroscience, 13(4), 1189-1215.
-
Nagatsu, T., Levitt, M., & Udenfriend, S. (1964). Tyrosine hydroxylase the initial step in norepinephrine biosynthesis. Journal of Biological Chemistry, 239(9), 2910-2917.
-
Levitt, M., Spector, S., Sjoerdsma, A., & Udenfriend, S. (1965). Elucidation of the rate-limiting step in norepinephrine biosynthesis in the perfused guinea-pig heart. Journal of Pharmacology and Experimental Therapeutics, 148(1), 1-8.
- Weiner N, Molinoff PB. Cathecolamines. (1989) En: Siegel GJ, Agranoff B, Albers RW, Molinoff PB eds. Basic Neurochemistry. 4th Ed. New York: Raven Press;. p. 233-51.
- Receptor and ion channel nomenclature. Trends Pharmacol Sci 1998.
-
Raiteri, M., Marchi, M., & Maura, G. (1982). Presynaptic muscarinic receptors increase striatal dopamine release evoked by ‘quasi-physiological’depolarization. European journal of pharmacology, 83(1-2), 127-129.
-
Feldman, R. S., Meyer, J. S., Quenzer, L. F., & Cooper, J. R. (1997). Principles of neuropsychopharmacology (No. 615+ 616 FEL). Sunderland: Sinauer Associates.
-
Humphrey, P. P. (1997). The characterization and classification of neurotransmitter receptors. Annals of the New York Academy of Sciences, 812(1), 1-13.
-
Kupfermann, I., Kandel, E. R., Schwartz, J. H., & Jessel, T. M. (1991). Principles of neural science.
Este texto se ofrece únicamente con propósitos informativos y no reemplaza la consulta con un profesional. Ante dudas, consulta a tu especialista.







