Duchenne Muscular Dystrophy


Escrito y verificado por el biólogo Samuel Antonio Sánchez Amador
Duchenne muscular dystrophy, or DMD, is a hereditary disease with a recessive inheritance pattern. In general terms, this disease is characterized by rapid and progressive muscle weakness that leads to the premature death of the patient.
According to epidemiological studies, this serious disorder occurs in 7.1 men and 2.8 women per 100,000 inhabitants. As low as this figure may seem, the global prevalence indicates that it’s the most frequent and severe neuromuscular disease that can present during childhood.
For this reason, knowing the characteristics and peculiarities of Duchenne muscular dystrophy is essential. Today, we’ll present everything you need to know about it, based on medical literature and scientific studies. Don’t miss the following article.
What’s Duchenne muscular dystrophy?
As we’ve said, DMD is an inherited disease marked by progressive muscle deficiency that gets worse very quickly. According to medical sources, the direct cause of this event is a degeneration of smooth, skeletal, and cardiac muscles. Let’s see why.
The role of dystrophin
Dystrophin is a cytoskeletal protein whose function is to prevent damage to the membrane of muscle cells —known as sarcolemma— during the process of muscle contraction. Here’s a series of facts about dystrophin that will help us understand Duchenne muscular dystrophy:
- It’s a muscle structural protein encoded by the DMD gene. It’s the largest protein encoded in the human genome.
- It acts as a shock absorber at the tissue level.
- It binds to the muscle membrane and helps maintain the structure and organization of the cells that make it up.
- Without dystrophin, muscles suffer progressive damage that eventually leads to the death of tissue.
Specialized portals, such as Duchenne Parent Project Spain, delve a little deeper into this subject. According to their fact sheets, damage to muscle cell membranes is a bigger problem, as substances flow in and out of these structures abnormally.
For example, the entry of calcium ions into muscle cells and fibers —mainly responsible for contraction—, in the absence of dystrophin, can cause the release of harmful enzymes and compounds that cause tissue damage.
The effect of the lack of dystrophin
Excessive entry of calcium ions causes membrane rupture, allowing more extracellular calcium to enter the cell down the gradient. This cascading reaction is expected to further promote muscle breakdown.
Ultimately, the destroyed muscle cells are replaced by fibrous connective tissue and adipose tissue, causing a loss of motor function in the patient. These spaces restrict the process of muscle contraction, leading to stiffness and impaired muscle functionality. In the absence of dystrophin, the number of muscle cells is reduced, and those that remain lose their capacity.
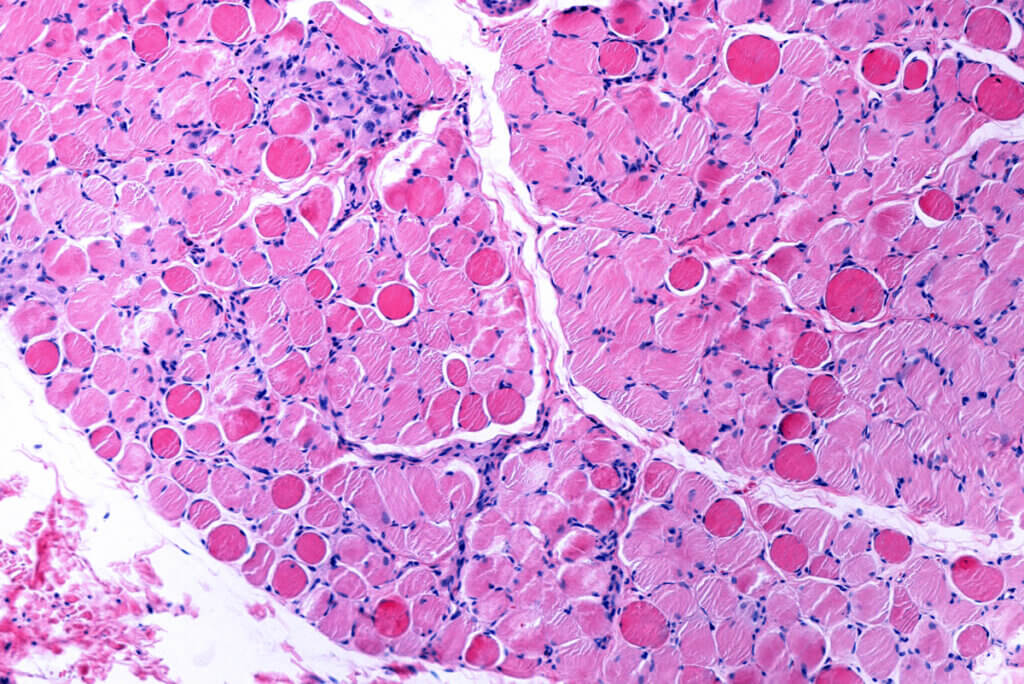
The causes of DMD
As we’ve said, DMD is a recessive genetic disorder. An allele that’s only expressed in the presence of another allele that’s equal to it is considered recessive. Let’s give an example:
- The R allele codes for brown eye color and is dominant in nature.
- The r allele codes for blue eye color and is recessive.
We must bear in mind that an allele is each one of the alternative forms of a gene and that —in typical genetics— an individual inherits in pairs, one from the father and one from the mother. So, the possible combinations in this example would be RR, Rr, and rr.
According to the aforementioned definition, blue eyes would only be expressed in the rr genotype. In the rest of the cases, the brown allele would mask the action of the blue allele, as the brown allele is dominant. With this fictitious example, we can understand a bit about the inheritance of Duchenne muscular dystrophy.
A genetic disease
In this case, we’re dealing with a recessive disease linked to the X chromosome. The gene that encodes dystrophin is located on the short arm of the X chromosome, more specifically in the Xp21 locus. It’s a large gene, as it has more than 2.5 million base pairs and 79 coding regions.
Two-thirds of DMD cases are associated with duplication or deletion of certain segments within this gene, while the remaining percentage is due to point mutations that are too small to be detected. This of course has a negative effect on the process of DNA transcription.
Without going too far into the matter, we’ll say that under normal conditions, messenger RNA reads the cell’s DNA and genes and carries the information to the ribosomes, where the protein encoded in the genome is synthesized. In this disorder, the reading frame that encodes dystrophin is altered, making its synthesis impossible.
According to professional genetic databases, the statistics of the causes of the disease are distributed as follows:
- 60-70% of patients show deletions in the DMD gene. This means that certain sections of the coding genetic material have been removed as a result of a mutation.
- 10% of patients show duplications, that is, repetitions of genetic information within the DMD gene.
- The remaining 20% of cases are due to small errors.
Due to genetic patterns of inheritance, men are much more likely to develop DMD than women.
The symptoms of Duchenne muscular dystrophy
Once we’ve clarified the causes of the disease, the next step is to describe the symptoms of Duchenne muscular dystrophy. According to the United States National Library of Medicine, these first signs appear before the patient is six years old and can even appear during the lactation period.
Among the symptoms of the disease, we find the following:
- Fatigue.
- Learning problems and intellectual disability.
- Muscle weakness: When running and jumping, frequent falls, difficulty getting up from a certain position, and breathing problems that worsen.
- Progressive difficulty walking: This begins around 12 years of age and associated heart disease begins to manifest itself around 20 years of life.
In general, the diagnosis can be delayed until 3-5 years of age due to the lack of distinguishable signs, but at 12, the infant usually requires a wheelchair to move. Unfortunately, the average life expectancy of patients is 30 years.
Diagnosis
For the diagnosis of this disease, a complete examination of the nervous system, lungs, heart, and muscles is required. Some tests to perform are:
- Electromyography: Graphic recording technique of the electrical activity presented by the muscles.
- DNA tests: Allow the detection of the specific mutation in most cases of DMD.
- Muscle biopsy: A small segment of muscle tissue may be obtained to look for the protein dystrophin. If it is absent, the diagnosis is clear.
A patient with Duchenne muscular dystrophy will show muscle wasting, contractures, deformities, cardiac abnormalities, and respiratory disorders. All these symptoms can also indicate to the professional that the general picture is caused by DMD.

The treatment of Duchenne muscular dystrophy
Until 2017, only palliative treatment was proposed to address Duchenne muscular dystrophy. That is, providing the best possible existence to the patient until the disease caused their death.
This may include assisted ventilation to help the infant breathe, drugs to stabilize heart activity, orthotics, and some specific surgeries to prevent the complete loss of mobility. Unfortunately, it’s still a disorder in which death is only a matter of time.
These measures can also be supplemented with steroids, proteins, and vitamins; substances administered to the patient in order to slow down the rapid muscular dystrophy that characterizes the disease. The effects of these therapies aren’t entirely effective.
Still, in recent times, gene therapy has begun to be used to treat patients with DMD. This disease has already been cured in mice, dogs, and cats and is in the experimental phase in humans. Of course, the future of this treatment is promising.
Hope for the future regarding Duchenne muscular dystrophy
As we’ve been able to see in these lines, Duchenne’s disease is a rapidly progressing, aggressive genetic disorder with a very low survival rate —almost all patients die before the age of 30. Muscular dystrophy progresses and the patient usually dies from cardiac or respiratory complications.
Despite the data presented here, there’s a ray of hope, as gene therapy is reporting very promising results under experimental conditions. Let’s hope it’s just a matter of time.
Duchenne muscular dystrophy, or DMD, is a hereditary disease with a recessive inheritance pattern. In general terms, this disease is characterized by rapid and progressive muscle weakness that leads to the premature death of the patient.
According to epidemiological studies, this serious disorder occurs in 7.1 men and 2.8 women per 100,000 inhabitants. As low as this figure may seem, the global prevalence indicates that it’s the most frequent and severe neuromuscular disease that can present during childhood.
For this reason, knowing the characteristics and peculiarities of Duchenne muscular dystrophy is essential. Today, we’ll present everything you need to know about it, based on medical literature and scientific studies. Don’t miss the following article.
What’s Duchenne muscular dystrophy?
As we’ve said, DMD is an inherited disease marked by progressive muscle deficiency that gets worse very quickly. According to medical sources, the direct cause of this event is a degeneration of smooth, skeletal, and cardiac muscles. Let’s see why.
The role of dystrophin
Dystrophin is a cytoskeletal protein whose function is to prevent damage to the membrane of muscle cells —known as sarcolemma— during the process of muscle contraction. Here’s a series of facts about dystrophin that will help us understand Duchenne muscular dystrophy:
- It’s a muscle structural protein encoded by the DMD gene. It’s the largest protein encoded in the human genome.
- It acts as a shock absorber at the tissue level.
- It binds to the muscle membrane and helps maintain the structure and organization of the cells that make it up.
- Without dystrophin, muscles suffer progressive damage that eventually leads to the death of tissue.
Specialized portals, such as Duchenne Parent Project Spain, delve a little deeper into this subject. According to their fact sheets, damage to muscle cell membranes is a bigger problem, as substances flow in and out of these structures abnormally.
For example, the entry of calcium ions into muscle cells and fibers —mainly responsible for contraction—, in the absence of dystrophin, can cause the release of harmful enzymes and compounds that cause tissue damage.
The effect of the lack of dystrophin
Excessive entry of calcium ions causes membrane rupture, allowing more extracellular calcium to enter the cell down the gradient. This cascading reaction is expected to further promote muscle breakdown.
Ultimately, the destroyed muscle cells are replaced by fibrous connective tissue and adipose tissue, causing a loss of motor function in the patient. These spaces restrict the process of muscle contraction, leading to stiffness and impaired muscle functionality. In the absence of dystrophin, the number of muscle cells is reduced, and those that remain lose their capacity.

The causes of DMD
As we’ve said, DMD is a recessive genetic disorder. An allele that’s only expressed in the presence of another allele that’s equal to it is considered recessive. Let’s give an example:
- The R allele codes for brown eye color and is dominant in nature.
- The r allele codes for blue eye color and is recessive.
We must bear in mind that an allele is each one of the alternative forms of a gene and that —in typical genetics— an individual inherits in pairs, one from the father and one from the mother. So, the possible combinations in this example would be RR, Rr, and rr.
According to the aforementioned definition, blue eyes would only be expressed in the rr genotype. In the rest of the cases, the brown allele would mask the action of the blue allele, as the brown allele is dominant. With this fictitious example, we can understand a bit about the inheritance of Duchenne muscular dystrophy.
A genetic disease
In this case, we’re dealing with a recessive disease linked to the X chromosome. The gene that encodes dystrophin is located on the short arm of the X chromosome, more specifically in the Xp21 locus. It’s a large gene, as it has more than 2.5 million base pairs and 79 coding regions.
Two-thirds of DMD cases are associated with duplication or deletion of certain segments within this gene, while the remaining percentage is due to point mutations that are too small to be detected. This of course has a negative effect on the process of DNA transcription.
Without going too far into the matter, we’ll say that under normal conditions, messenger RNA reads the cell’s DNA and genes and carries the information to the ribosomes, where the protein encoded in the genome is synthesized. In this disorder, the reading frame that encodes dystrophin is altered, making its synthesis impossible.
According to professional genetic databases, the statistics of the causes of the disease are distributed as follows:
- 60-70% of patients show deletions in the DMD gene. This means that certain sections of the coding genetic material have been removed as a result of a mutation.
- 10% of patients show duplications, that is, repetitions of genetic information within the DMD gene.
- The remaining 20% of cases are due to small errors.
Due to genetic patterns of inheritance, men are much more likely to develop DMD than women.
The symptoms of Duchenne muscular dystrophy
Once we’ve clarified the causes of the disease, the next step is to describe the symptoms of Duchenne muscular dystrophy. According to the United States National Library of Medicine, these first signs appear before the patient is six years old and can even appear during the lactation period.
Among the symptoms of the disease, we find the following:
- Fatigue.
- Learning problems and intellectual disability.
- Muscle weakness: When running and jumping, frequent falls, difficulty getting up from a certain position, and breathing problems that worsen.
- Progressive difficulty walking: This begins around 12 years of age and associated heart disease begins to manifest itself around 20 years of life.
In general, the diagnosis can be delayed until 3-5 years of age due to the lack of distinguishable signs, but at 12, the infant usually requires a wheelchair to move. Unfortunately, the average life expectancy of patients is 30 years.
Diagnosis
For the diagnosis of this disease, a complete examination of the nervous system, lungs, heart, and muscles is required. Some tests to perform are:
- Electromyography: Graphic recording technique of the electrical activity presented by the muscles.
- DNA tests: Allow the detection of the specific mutation in most cases of DMD.
- Muscle biopsy: A small segment of muscle tissue may be obtained to look for the protein dystrophin. If it is absent, the diagnosis is clear.
A patient with Duchenne muscular dystrophy will show muscle wasting, contractures, deformities, cardiac abnormalities, and respiratory disorders. All these symptoms can also indicate to the professional that the general picture is caused by DMD.

The treatment of Duchenne muscular dystrophy
Until 2017, only palliative treatment was proposed to address Duchenne muscular dystrophy. That is, providing the best possible existence to the patient until the disease caused their death.
This may include assisted ventilation to help the infant breathe, drugs to stabilize heart activity, orthotics, and some specific surgeries to prevent the complete loss of mobility. Unfortunately, it’s still a disorder in which death is only a matter of time.
These measures can also be supplemented with steroids, proteins, and vitamins; substances administered to the patient in order to slow down the rapid muscular dystrophy that characterizes the disease. The effects of these therapies aren’t entirely effective.
Still, in recent times, gene therapy has begun to be used to treat patients with DMD. This disease has already been cured in mice, dogs, and cats and is in the experimental phase in humans. Of course, the future of this treatment is promising.
Hope for the future regarding Duchenne muscular dystrophy
As we’ve been able to see in these lines, Duchenne’s disease is a rapidly progressing, aggressive genetic disorder with a very low survival rate —almost all patients die before the age of 30. Muscular dystrophy progresses and the patient usually dies from cardiac or respiratory complications.
Despite the data presented here, there’s a ray of hope, as gene therapy is reporting very promising results under experimental conditions. Let’s hope it’s just a matter of time.
- Researchers Try to Update Prevalence of Duchenne Muscular Dystrophy, AJMC. Recogido a 1 de noviembre en https://www.ajmc.com/view/researchers-try-to-update-prevalence-of-duchenne-muscular-dystrophy
- Distrofia muscular de Duchenne, orphanet.org. Recogido a 1 de noviembre en https://www.orpha.net/consor/www/cgi-bin/OC_Exp.php?lng=ES&Expert=98896
- La proteína distrofina, Duchenne Parent Project España. Recogido a 1 de noviembre en https://www.duchenne-spain.org/la-distrofina/
- Delección, NIH. Recogido a 1 de noviembre en https://www.genome.gov/es/genetics-glossary/Delecion#:~:text=Una%20deleci%C3%B3n%20es%20un%20tipo,todo%20un%20fragmento%20de%20cromosoma.
- Homo sapiens dystrophin (DMD), RefSeqGene (LRG_199) on chromosome X, NCBI. Recogido a 1 de noviembre en https://www.ncbi.nlm.nih.gov/nuccore/256355061
- Distrofia muscular de Duchenne, Medlineplus.gov. Recogido a 1 de noviembre en https://medlineplus.gov/spanish/ency/article/000705.htm
- Bouwles, DE; McPhee SW, Li C, Gray SJ, Samulski JJ, Camp AS, Li J, Wang B, Monahan PE, Rabinowitz JE, Grieger JC, Govindasamy L, Agbandje-McKenna M, Xiao X, Samulski RJ. (febrero de 2012). «Phase 1 gene therapy for Duchenne muscular dystrophy using a translational optimized AAV vector.».
- Guiraud, Simon, and Kay E. Davies. “Pharmacological advances for treatment in Duchenne muscular dystrophy.” Current opinion in pharmacology 34 (2017): 36-48.
Este texto se ofrece únicamente con propósitos informativos y no reemplaza la consulta con un profesional. Ante dudas, consulta a tu especialista.







